 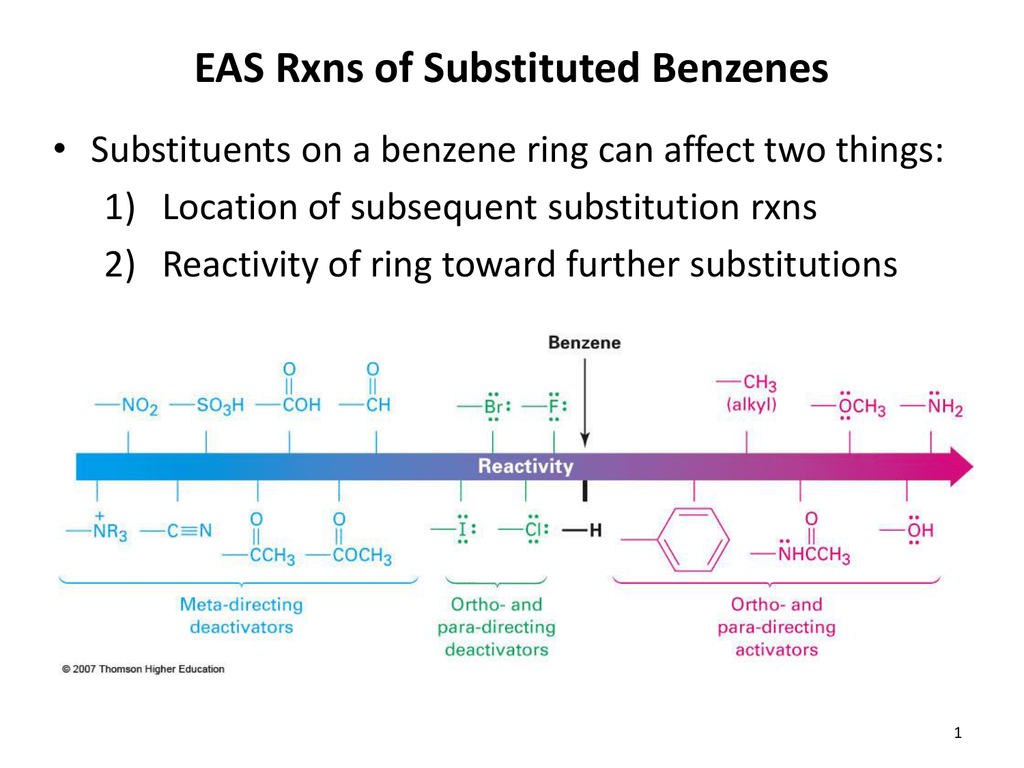
Instead of using numbers to indicate substituents on a benzene ring, ortho- (o-), meta- (m-), or para (p-) can be used in place of positional markers when there are two substituents on the benzene ring (disubstituted benzenes). Therefore, it's the weakest acid of all under investigation. Naming Benzene molecules with two substituents:Right beside each other (1,2) Ortho (o-)One carbon separation (1,3) Meta (m-)Two carbon separation (1,4). Ortho-, Meta-, Para- (OMP) Nomenclature for Disubstituted Benzenes. As in other posts I have seen and learnt that, if one is given four compounds, namely para-nitrophenol, ortho-nitrophenol, meta-nitrophenol and phenol and is told to arrange them in order of acidity, then one has to take into account the $\text$ effect (a deactivating group). But in general I would say that para- products are produced in greater quantity over the ortho- products becuase of the steric hindrance of the isopropyl group like you mentioned. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
